Sodium Hyaluronate
Sodium Hyaluronate Specification
- Storage Instructions
- Store in cool, dry place, protected from light
- Usage Type
- External use, oral formulations
- Function
- Moisturizing, viscoelastic agent
- Shelf Life
- 24 months
- Accuracy
- High Purity ( 98%)
- Measurement Range
- Depends on end-use application
- Features
- High Purity, Moisture-retaining, Biocompatible
- Equipment Type
- Pharmaceutical Raw Material
- Material
- Sodium Hyaluronate
- Condition
- New
- Technology
- Biotechnological Fermentation
- Portable
- Yes
- Operating Type
- Manual
- Use
- Used in cosmetics, pharmaceuticals, and medical formulations
- Weight
- As per packaging, generally 1 kg/bag or customized
- Color
- White or off-white powder
- Assay (Sodium Hyaluronate)
- 98.0%
- Heavy Metals
- 10 ppm
- Loss on Drying
- 10.0%
- Synonyms
- Hyaluronic Acid Sodium Salt
- CAS Number
- 9067-32-7
- Grade
- Cosmetic and Pharmaceutical Grade
- Application
- Skin care, injectable solutions, ophthalmic products
- Molecular Weight
- 8000-2000000 Da (customizable)
- Appearance
- White to off-white powder
- Packing
- Aluminum foil bag/drum
- Microbial Limit
- Meets pharmacopeial standards
- Endotoxin Level
- < 0.5 EU/mg
- Solubility
- Freely soluble in water
- pH value (1% solution)
- 6.0-7.5
Sodium Hyaluronate Trade Information
- Minimum Order Quantity
- 1 Unit
- Supply Ability
- 5000 Units, , Per Day
- Delivery Time
- 7-20 Days
About Sodium Hyaluronate
We are an illustrious organization, successfully engaged in providing the best quality of Sodium Hyaluronate .The offered ion is manufactured with the use of optimum qualityraw materials under the supervision of ingenious professionals.The provided solution is widely used in ophthalmic treatments in order to relieve soreness and dryness of eyes.In addition, our clients can purchase this Sodium Hyaluronate .
Key points:
- Accurate composition and known for side-effects free nature
- Non-hazardous and highly effective
- Known for excellent results
- Packed hygienically and is known for its longer shelf life
Description: Sodium Hyaluronate is a sterile, non-pyrogenic, viscolastic planning of profoundly filtered, no incendiary, of Sodium Hyaluronate Ophthalmic Solution. IO-LON contains 18 mg/ml. of sodium Hyaluronate broke up in physiological sodium chloride phosphate support (pH 6.8-7.4)
Qualities: Sodium hyaluronate is gotten from bacterial aging. Its consistency is in charge of upkeep of space in the foremost chamber and permits the control of tissue. Amid the operation without getting away trough the entry point.
The surgical shear rate thickness decides the imperviousness to the instruments and embeds. It has no give free development to the instruments and simple passage for the insert into the expanded capsular pack.
Moreover the 1.8% arrangement of Sodium Hyaluronate is straightforward. It ensures corneal endothelial cell and other visual structures. Sodium Hyaluronate does not meddle with epithelization and typical injury recuperating.
Outstanding Purity & Versatile Applications
With an assay of 98%, our Sodium Hyaluronate offers unmatched purity for diverse uses in cosmetic and pharmaceutical formulations. Its moisture-binding and viscoelastic characteristics make it ideal for creams, injectable therapies, and ophthalmic solutions. Consistent particle quality and adaptable molecular weight ensure compatibility with every application.
Stringent Quality Assurance
Every batch adheres to rigorous pharmacopeial standards, including <0.5 EU/mg endotoxins, 10 ppm heavy metals, and a loss on drying of 10%. Advanced biotechnological fermentation guarantees high biocompatibility, making it suitable for external, oral, and injectable forms, assuring safety and efficacy in finished products.
User-Friendly Packaging & Storage
Our Sodium Hyaluronate comes in robust aluminum foil bags or drums, designed for portability and easy use. Packaging is typically 1 kg per bag, with customization available. For maximum shelf life, store in a cool, dry environment, away from direct light, ensuring quality is maintained throughout its 24-month shelf life.
FAQs of Sodium Hyaluronate:
Q: How is Sodium Hyaluronate typically used in cosmetic and pharmaceutical products?
A: Sodium Hyaluronate serves as a key ingredient in moisturizing skin care creams, serums, injectable fillers, and ophthalmic solutions due to its exceptional moisture retention and viscoelasticity. Its biocompatibility makes it suitable for both topical and injectable formulations, adapting to various cosmetic and medical use cases.Q: What benefits does Sodium Hyaluronate offer for skin and medical applications?
A: This ingredient hydrates deeply, aids skin elasticity, and helps smooth fine lines. In medical products, it acts as a viscoelastic agent for joint and eye therapies, promoting tissue healing and comfort while maintaining compatibility with human tissues.Q: Where should Sodium Hyaluronate be stored for optimal preservation?
A: To maintain its quality, Sodium Hyaluronate should be stored in a cool, dry place, protected from direct light, and kept in its original aluminum foil packaging. When stored properly, it remains stable and effective for up to 24 months.Q: What process is used to manufacture Sodium Hyaluronate?
A: Sodium Hyaluronate is produced through a controlled biotechnological fermentation process. This technology ensures high purity, consistent molecular weight, and low endotoxin levels, making it suitable for sensitive pharmaceutical and cosmetic applications.Q: When can Sodium Hyaluronate be incorporated into formulations?
A: It can be added during the formulation stage of both external and injectable products, provided that handling conditions preserve its purity and efficacy. It is compatible with water-based formulations and dissolves easily due to its high solubility.Q: How customizable is the molecular weight of Sodium Hyaluronate for different applications?
A: Molecular weight is tailor-made, ranging from 8,000 to 2,000,000 Da, depending on end-use. Lower molecular weights are typically used for greater absorption in serums, while higher weights offer enhanced viscoelastic properties for medical injectables.Q: What safety and quality standards does this Sodium Hyaluronate meet?
A: Each batch meets strict guidelines: assay 98%, endotoxin level <0.5 EU/mg, heavy metals 10 ppm, and microbial limits per pharmacopeial norms, ensuring it is safe for cosmetic, pharmaceutical, and medical formulations.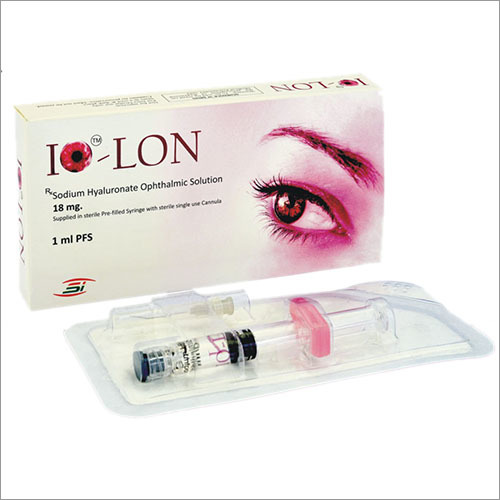

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Eye Care Products Category
Eye Shield
Minimum Order Quantity : 1 Unit
Wall Mounted : No
Operating Type : Manual
Weight : Approx. 20 g
Storage Instructions : Store in a cool, dry place away from direct sunlight
Portable : Yes
Eye Wipes
Minimum Order Quantity : 1 , , Unit
Wall Mounted : No
Operating Type : Manual
Weight : Lightweight
Storage Instructions : Store in a cool, dry place
Portable : Yes
Eye Drapes
Minimum Order Quantity : 1 Unit
Wall Mounted : No
Operating Type : Manual
Weight : Lightweight
Storage Instructions : Store in cool, dry place away from sunlight
Portable : Yes
Retinal Products (Silicone Band, Silicone Tire, Silicone Oil, Silicone Sponge)
Minimum Order Quantity : 1 , , Unit
Wall Mounted : No
Operating Type : Other, Manual / Surgical Assisted
Weight : Lightweight (varies by product type and size)
Storage Instructions : Store in a cool, dry place away from direct sunlight
Portable : Yes

 Send Inquiry
Send Inquiry





 Send Inquiry
Send Inquiry Send SMS
Send SMS