Ophthalmic Products
Ophthalmic Products Specification
- Equipment Type
- Ophthalmic Products
- Material
- Medical Grade Stainless Steel / High-quality Polymer
- Technology
- Precision Machined / Advanced Manufacturing Techniques
- Properties
- Anti-reflective, Scratch-resistant, Lightweight
- Grade
- Surgical Grade
- Lens Specification
- Aspheric, Hydrophobic Coating, Multi-focal options
- Lens Diameter
- 10 mm to 15 mm (varies by product)
- Prism
- Available on select diagnostic lenses
- Implants Specifications
- Biocompatible, UV Absorbing, Foldable
- Driven Method
- Manual and Motorized options
- Power Consumption
- Low (for powered devices), Typically below 15W
- Voltage
- 110V/220V (for electrical devices)
- Dimension (L*W*H)
- Varies by Product: (e.g., 12 x 6 x 4 cm)
- Weight
- 45 grams (average per piece)
- Usage
- Cataract Surgery, Retinal Procedures, Diagnostic Purposes
- Sterilized
- Pre-sterilized and Non-sterilized available
- Sterilization Mode
- ETO, Gamma, Autoclave
- Packaging
- Sterile Blister Pack / Medical Grade Pouch
- Shelf Life
- 3-5 Years from Manufacturing
- Safety Standard
- Complies with EN/IEC Safety Standards
- Application
- Hospital, Clinics, Surgical Centers
- Temperature Tolerance
- -10C to +55C
- Resistance
- Corrosion-resistant, Chemical-resistant
- Compatibility
- Universal Fit for Major Ophthalmic Instruments
- Color
- Clear, Blue, Green (for identification)
- Surface Finish
- Polished / Matte / Textured
Ophthalmic Products Trade Information
- Minimum Order Quantity
- 1 , , Unit
- Supply Ability
- 5000 Units Per Day
- Delivery Time
- 7-20 Days
About Ophthalmic Products
Having seven years of experience in this field, we are consistently engaged in providing the best quality Ophthalmic Products like Eye shield, absorbant sticks, Ophthalmic instruments. The offered products are manufactured with the aid of highly advanced techniques using superior quality basic materials. These products are used in treatment of retinal infections and several other problems related to eye injury. Mostly preferred by clients of medical industry, these products are put forth in different sizes, patterns and finishes. Additionally, we provide these Ophthalmic Products (Eye shield, absorbant sticks, Ophthalmic instruments) at reasonable prices to our clients.
Key points:
- Manufactured with high precision under hygienic conditions
- Supreme finish and known for high durability
- Easy and safe to use
- Biodegradable and non-hazardous to environment
Universal Compatibility and Safe Packaging
Our ophthalmic products are designed for a universal fit with major ophthalmic instruments, ensuring seamless integration and versatility in practice. Packaged in sterile blister packs or medical-grade pouches, each item is protected against contamination and environmental damage, supporting a shelf life of 3-5 years from the date of manufacturing.
Engineering Excellence and Durability
Crafted with precision-machined technology and advanced manufacturing techniques, these products are available in both medical-grade stainless steel and high-quality polymer. Surface finishespolished, matte, or texturedallow for tailored tactile experiences. All devices are corrosion-resistant, chemical-resistant, and compliant with stringent international safety standards.
Efficiency and Patient Safety in Clinical Use
Sterilized through ETO, Gamma, or Autoclave methods, these ophthalmic solutions ensure high standards of sterility for clinical settings. Both manual and motorized varieties are available, featuring low power consumption (typically below 15W) for powered devices. Additional features such as anti-reflective, hydrophobic coatingsand multifocal lenses on select itemssupport superior outcomes for patients.
FAQs of Ophthalmic Products:
Q: How are these ophthalmic products typically used in medical procedures?
A: These products are commonly utilized in cataract surgery, retinal procedures, and diagnostic examinations in hospitals, clinics, and surgical centers. Their universal design enables integration with most major ophthalmic instruments, making them suitable across diverse clinical applications.Q: What sterilization methods are available for these products?
A: You can choose between pre-sterilized and non-sterilized options. Sterilization is performed using ETO, gamma radiation, or autoclave, ensuring reliable infection control and user convenience based on clinical requirements.Q: When does the shelf life of the product start, and how long does it last?
A: The shelf life is calculated from the date of manufacturing and typically lasts 35 years, provided the packaging remains unopened or intact, maintaining the sterility and integrity of each product.Q: Where can these ophthalmic products be used?
A: These items are ideal for use in hospitals, specialized eye clinics, and surgical centers. As they are compatible with most major ophthalmic instruments and meet international safety standards, their deployment is versatile and widely accepted.Q: What are the main benefits of the materials and finishes used?
A: By using medical-grade stainless steel and high-quality polymers, the products resist corrosion and chemicals, supporting longevity and hygiene. Multiple finishespolished, matte, texturednot only enhance handling but also allow practitioners to choose based on preference and procedure type.Q: How does color coding of products help during procedures?
A: Clear, blue, and green color options facilitate quick identification and organization in the operating room or diagnostic suite. This system helps staff operate efficiently, minimizing confusion and potential errors during time-sensitive procedures.Q: What is the process for exporting and supplying these products from India?
A: As an established exporter, manufacturer, supplier, and trader in India, the company adheres to regulatory standards for medical devices. Products are packaged with proper documentation, ensuring compliance for international shipments and prompt delivery to worldwide customers.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in General Ophthalmic Products Category
Lissamine Strip
Minimum Order Quantity : 1 , , Unit
Grade : Medical
Technology : Other, Colorimetric staining
Properties : Sterile, individually packed
Material : Cellulose
Usage : For ocular surface staining, assessment of dry eye, and detection of devitalized ocular cells
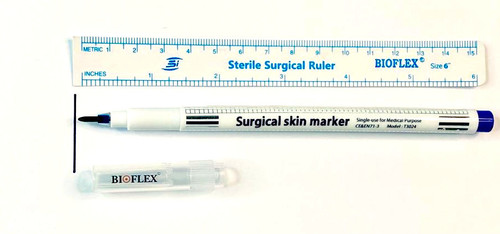
Surgical Skin Marker
Grade : Surgical Grade
Technology : Other, Manual marking pen
Properties : Disposable, sterile, nontoxic
Material : Medicalgrade plastic and nontoxic ink
Usage : Marking surgical sites on skin
Rose Bengal Strip
Minimum Order Quantity : 1 Unit
Grade : Medical grade
Technology : Other, Imbibitionbased dye release
Properties : Sterile, individually packed, rapid staining ability
Material : Plastic backing with medical filter paper impregnated with Rose Bengal dye
Usage : Ophthalmic surface staining, dry eye evaluation
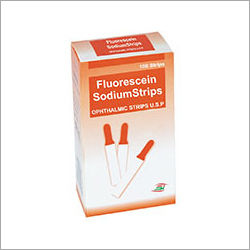
Fluorescein Sodium Strips
Minimum Order Quantity : 1 Unit
Grade : Medical Grade
Technology : Other, Colorimetric Diagnostic
Properties : Singleuse, Noninvasive, Biocompatible
Material : Cellulose Filter Paper
Usage : For corneal staining and tear film analysis in ophthalmic examinations

 Send Inquiry
Send Inquiry





 Send Inquiry
Send Inquiry Send SMS
Send SMS